Therefore, while the Law of Definite Proportions can be used to compare two experiments in which hydrogen and oxygen react to form water, the Law of Definite Proportions can not be used to compare one experiment in which hydrogen and oxygen react to form water, and another experiment in which hydrogen and oxygen react to form hydrogen peroxide (peroxide is another material that can be made from hydrogen and oxygen). 
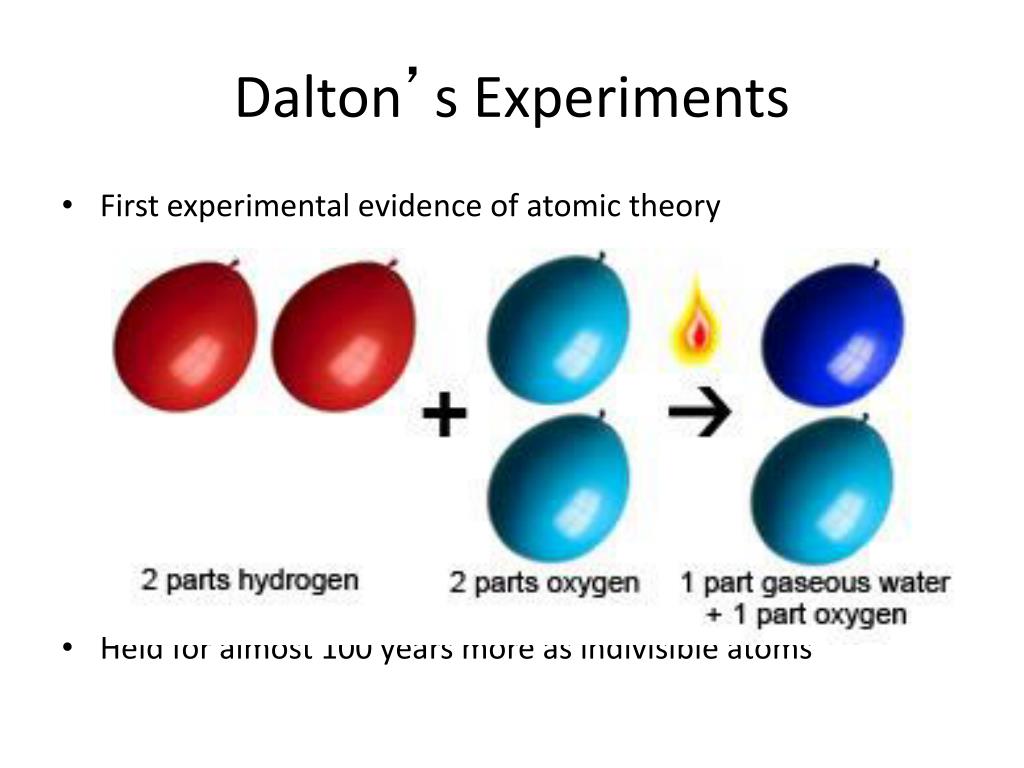
The Law of Definite Proportions applies when elements are reacted together to form the same product. A theory of chemical combination, first stated by John Dalton in 1803. Law of Definite Proportions states that in a given type of chemical substance, the elements are always combined in the same proportions by mass. Daltons atomic theory was a logical hypothesis on the idea of the issue and it expressed that all matter was comprised of little, unified particles known as atoms. Similarly, when 2 grams of A react with 16 grams of B, they must produce 18 grams of C. Dalton’s Atomic Theory: In the course of this research into gases, Dalton also discovered that certain gases could only be combined in certain proportions, even if two different compounds shared. ---teachoo-01-01.jpg)
The principle of conservation of mass was first. Historically, already the ancient Greeks proposed the idea that the total amount of matter in the universe is constant. If 1 gram of A reacts with 8 grams of B, then by the Law of Conservation of Mass, they must produce 9 grams of C. Near the end of the 18th century, two laws about chemical reactions emerged without referring to the notion of an atomic theory. \): If 1 gram of A reacts with 8 grams of B, then by the Law of Definite Proportions, 2 grams of A must react with 16 grams of B.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
